by Andrzej Kajetan Wróblewski
As with a number of scientific discoveries, Henri Becquerel’s discovery of uranium’s radioactivity occurred by accident. While investigating Wilhelm Conrad Röntgen’s recent work on X-rays, Becquerel decided to test Poincaré’s hypothesis that the emission of X-rays could be related to phosphorescence, essentially the delayed emission of light by a substance after its exposure to light. As he later said in his Nobel lecture (Becquerel 1903): “At the beginning of 1896, on the very day that news reached Paris of Röntgen’s experiments and of the extraordinary properties of the rays emitted by the phosphorescent walls of the Crookes tubes, I thought of carrying out research to see whether all phosphorescent material emitted similar rays. The results of the experiment did not justify this idea, but in this research I encountered an unexpected phenomenon.”
During the course of his research, Becquerel wrapped exposed uranium mineral in photographic plates and black material to prepare for an experiment requiring bright sunlight. However, since the weather in Paris had been overcast for days, he kept the little exposed mineral and the plates in a drawer awaiting for a sunny day. Once the weather improved, Becquerel decided to develop the plate and found, to his surprise, that it was blackened. At the meeting of the Academy of Science on 2 March 1896, he announced that the uranium mineral emitted unknown penetrating radiation by itself (Becquerel 1896a).
 |
Wilhelm Conrad Röntgen
(1845–1923). |
After this breakthrough, Becquerel began studying the newly discovered radiation in more detail. He presented his results at three meetings of the Academy of Science in March 1896. On 9 March, he announced that the rays emitted by the double sulphate of uranium and potassium were capable of discharging an electroscope after passing through a 2-millimetre-thick aluminium plate. He also found that the invisible rays could be reflected and refracted (Becquerel 1896b). On 23 March, he presented more detailed results on the ionizing power of the new rays. Using a gold leaf electroscope, Becquerel compared the rate of discharge (radiation) of a potassium uranyl sulphate crystal with a Crookes’ tube and found that the effect from the tube was over 100 times greater than that of the crystal (Becquerel 1896c). On 30 March, Becquerel announced (Becquerel 1896d) that the rays emitted by uranium salts were doubly refracted by tourmaline, whereas in a parallel experiment with a Crookes’ tube no such effect was detected for the cathode rays.
At the five meetings of the Academy of Sciences in March 1896 there were more than 30 reports on X-rays. Amidst this flood of reports, the communications by Becquerel on uranium radiation didn’t cause much excitement and the initial interest in the new rays faded rapidly. There was a proliferation of false or doubtful observations of radiation similar to uranic rays in a variety of substances, and yet these results were unreliable due to the relatively poor quality of the photographic plate. To other leading scientists at the time, the uranium rays appeared to have “normal” properties, similar to those of ordinary light, and were therefore regarded as less intriguing than the mysterious X-rays.
Thus, when John Joseph Thomson delivered the Rede Lecture on “The Röntgen Rays” at Cambridge University on 10 June 1896, he had this to say (Thomson 1896):
“Since the discovery of the Röntgen rays, Becquerel has discovered a new kind of light, which in its properties resembles the Röntgen rays more closely than any kind of light hitherto known. . . . Becquerel has shown that the radiation from the uranium salts can be polarized, so that it is undoubtedly light: it can also be refracted. It forms a link between the Röntgen rays and ordinary light, it resembles the Röntgen rays in its photographic action, in power of penetrating substances opaque to ordinary light, and in the characteristic electrical effect, while it resembles ordinary light in its capacity for polarisation, in its liability to refraction.”
Other physicists were of a similar opinion. For example, Oscar M. Stewart of Cornell University had this to say about the rays in a review published in April 1898 (Stewart 1898):
“Becquerel rays occupy a unique position, inasmuch as far more is definitely known about them than any of the other ‘new’ “rays.” With X-rays nothing has been proven one way or the other about their character, save that if they are ultra-violet rays their wave-length must be extremely small, so small that the refractive index for nearly all bodies is practically unity. With the rays of Becquerel there can be no reasonable doubt that they are short transverse ether waves.”
Meanwhile, in August 1896, Pieter Zeeman of Leyden University discovered splitting of spectral lines in the magnetic field. Many physicists concentrated their attention on this long awaited connection between magnetism and light. It was around this time that Becquerel also left the “non-interesting” field of radioactivity, and from 1897 to 1899, he delivered at meetings of the Academy of Sciences, a number of papers on the Zeeman effect and the Faraday effect.
It is difficult to say how history would have been shaped if it were not for Maria Skłodowska-Curie who decided at the end of 1897 to study the “non-interesting” subject of uranium radiation. If she had continued her applied research on the magnetism of tempered steel, her name would probably not be widely known today.
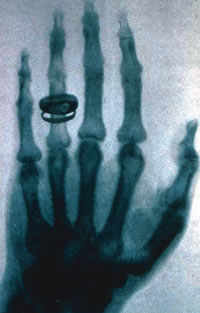 |
| For some time before Nov-Dec 1895, scientists had been reporting bizarre apparitions when they electrified the thin gas in vacuum tubes. On the Sunday before Christmas 1895, Wilhelm Conrad Röntgen invited his wife Bertha into the laboratory and took a shadow-graph of the bones of her hand with her wedding ring clearly visible. This is one of the most famous images in photographic history and propelled Röntgen in no time into international celebrity. The medical implications were immediately realized and the first images of fractured bones were being made by January 1896 even though none yet knew what the mystery rays were. The radiograph reproduced here is of the hand of Albert von Kolliker, made at the conclusion of Roentgen’s lecture and demonstration at the Wurzburg Physical-Medical Society on 23 January 1896. (Credit: AIP Emilio Segre Visual Archives, Lande Collection) |
Her first study of radioactivity (Skłodowska-Curie, 1898)—the term she first proposed—was a real break with the past. First, she applied a precise and sensitive electrometer; method much more reliable than the photographic method that gave qualitative, non repeatable, and often erroneous results because of the quality of the manufactured plates. Second, she decided to perform a systematic study of all available minerals, rocks, and other substances. This quickly resulted in a breakthrough since it was found that the intensity of radiation from various uranium minerals was not proportional to the amount of uranium they contained. This led Curie to hypothesize on the existence of a new unknown radioactive element. Her systematic studies led her to discover the radioactivity of thorium, which was also discovered independently by German physicist Gerhard Schmidt (Schmidt, 1898), who used a photographic method similar to that of Becquerel and found that thorium rays could be refracted and reflected (diffused) but not polarized.
Here is an excerpt from Curie’s paper (M. Curie 1898):
“I have examined a great number of metals, salts, oxides, and minerals. . . . All the compounds of uranium studied are very active and they are, in general, the more active the more uranium they contain. The compounds of thorium are very active. The oxide of thorium even exceeds metallic uranium in activity. It should be noted that two most active elements, uranium and thorium, are those which have the greatest atomic weight. . . . Two ores of uranium, pitchblende (uranium oxide), and chalcolite (phosphate of copper and uranium) are much more active than uranium itself. This fact is very remarkable and leads to the belief that these minerals may contain an element much more active than uranium. . . . To interpret the spontaneous radiation of uranium and thorium one might imagine that all space is constantly traversed by rays analogous to Röntgen rays but much more penetrating and able to be absorbed only by certain elements of high atomic weight, such as uranium and thorium.”
Thus, it was Curie’s first paper, published in April 1898, which again concentrated the interest of researchers on Becquerel rays. “It appeared that the results of my work were so interesting that Pierre Curie put aside his current research and joined me in the effort to extract and study new radioactive substances,” she wrote later in the introduction to her doctoral dissertation (M. Curie, 1903).
In July 1898, Maria and Pierre Curie announced the discovery of a new radioactive element (P. Curie and M. Curie 1898):
“Certain minerals containing uranium and thorium (pitchblende, chalcolite, uranite) are very active from the point of view of emission of Becquerel rays. In earlier work, one of us has shown that their activity is even greater than that of uranium and thorium, and has made the statement that this effect must be due to some other very active substance contained in a very small quantity in these minerals. . . . We believe, therefore, that the substance, which we have recovered from pitchblende contains a metal not yet described, related to bismuth in its analytical properties. If the existence of this new metal is confirmed, we propose to call it polonium, after the native country of one of us.”
However, because of the previous erroneous results by Becquerel, many physicists received the news about the new radioactive element with scepticism.
The January 1899 issue of Philosophical Magazine carried a paper by Ernest Rutherford (Rutherford 1899) that had been sent from Cambridge to the editors on 1 September 1898. Thus, it seems certain that Rutherford began studying radioactivity much before that date, probably at the same time as Curie. In the beginning of his paper, Rutherford stated that the following:
“The results of Becquerel showed that Röntgen and uranium radiations were very similar in their power of penetrating solid bodies and producing conduction in a gas exposed to them; but there was an essential difference between the two types of radiation. He found that uranium radiation could be refracted and polarised, while no definite results showing polarisation or refraction have been obtained for Röntgen radiation.”
 |
Henri Becquerel
(1852–1908). |
In his paper, Rutherford reported the important finding that uranium radiation contained two components differing in their penetrating power: strongly absorbed alpha radiation and penetrating beta radiation. It convinced Rutherford that uranium radiation is more complicated than it appeared from the study by Becquerel. Therefore, he questioned whether it was indeed necessary to postulate the existence of new substances:
“It is possible that the apparently very powerful radiation obtained from pitchblende by Curie may be partly due to the very fine state of division of the substance rather than to the presence of a new and powerful radiating substance.”
Meanwhile, Marie and Pierre Curie and Gustave Bémont continued their efforts to extract yet another substance from the pitchblende. The discovery of radium was announced on 26 December 1898 (Curie P., Curie M., Bémont G., 1898):
“The different reasons which we have enumerated lead us to believe that the new radio-active substance contains a new element to which we propose to give the name of radium . . . . The new radio-active substance certainly contains a very great proportion of barium; in spite of that, the radioactivity is considerable. The radio-activity of radium must therefore be enormous . . .”
 |
| Marie Curie in her laboratory. |
The discoveries of polonium and radium dispersed earlier doubts concerning the existence of new elements. In addition it convinced many physicists that radioactivity was an exciting field of study. Becquerel also returned to his research on uranium, and on 27 March 1899, he presented a paper to the Academy of Sciences. He stated that the intensity of the uranium radiation, as measured by its photographic action, appeared to be unchanged since May 1896; he also announced that the rays did not appear to be capable of refraction and polarization. All attempts to repeat two earlier experiments had failed. Thus, Becquerel withdrew the results that had contributed to the lack of interest in the field.
The following years were full of new discoveries. André Debierne (Debierne 1900) discovered actinium (results presented to the Academy of Sciences on 16 October 1899). Ernest Rutherford made an important impact on the study of radioactivity with the discovery of thorium emanation (1900) and the first theory of radioactive transmutations developed jointly with Frederick Soddy. In 1903, Becquerel and the Curies received the Nobel Prize in Physics.
It is difficult not to agree with the American historian Lawrence Badash who had this to say about the first years of radioactivity (Badash 1965): “In early 1898, radioactivity was something of a “dead horse”—it was there, but no one knew what to do with it. It took not only the discovery of thorium’s activity, first by Gerhard C. Schmidt and then by Marie Curie, but the subsequent discoveries of polonium and radium by the Curies to produce a sustained renewal of interest. For then it became apparent that this was an atomic phenomenon of great significance.” Andrzej Kajetan Wróblewski is professor emeritus in the Physics Department, University of Warsaw, formerly dean of the Physics Department (1986–1989) and rector of Warsaw University (1989–1993). His fields of interest include experimental physics of elementary particles and history of physics.
See the References section for works cited in this article.
Page
last modified 10 January 2011.
Copyright © 2003-2011 International Union of Pure and Applied Chemistry.
Questions regarding the website, please contact [email protected] |