|
Chemical
Education International, Vol. 5, No. 1, AN-1, Received June 15,
2004
A
project to disseminate a series of "Introduction to Chemistry"
by the Internet
by
Yoshito Takeuchi
Department of Chemistry
Faculty of Science
Kanagawa University
2946 Tsuchiya, Hiratsuka, Japan 259-1293
A
group of Japanese chemists launched a project to disseminate a
series of "Introduction to Chemistry" by the Internet
[1]. As the representative of the group, the author wishes to
introduce the purpose of this project, and the structure of the
series, to the potential users of the textbooks.
We wish the series would be used by many young students and citizens
who have some difficulty in purchasing textbooks though they are
very much interested in chemistry.
Preface
to "Introductory Chemistry"
Chemistry is a branch of science in which the kingdom of matter,
that is, composed of atoms, molecules, ions, and great numbers of
materials made of these, is studied in terms of molecules. It must
be added that chemistry also covers the kingdom of life in the sense
that even human being is composed of molecules. In chemistry, the
properties of materials, the laws that govern the kingdom of matter
and life, and the reactions taking place in nature and in laboratory
are studied. Furthermore, chemistry is a core part of novel and
frontier area of modern science/technology, i.e., biotechnology,
electronics, novel and functional materials. In other word, chemistry
is not only the core of basic science and technology, but also the
core of medical, pharmaceutical, agricultural environmental sciences,
or applied science/technology.
Because
of its pantoscopic nature, chemistry has many characteristics. Chemistry
treats a great number of materials, hence knowledge of chemistry
is accumulating . Knowledge of chemistry is never become obsolete
but simply accumulating. Knowledge obtained in the past is still
important in modern era.
The
methods employed by chemistry to investigate materials are also
accumulating. Side by side with modern techniques such as spectroscopy
and quantum chemical calculations, traditional methods such as melting/boiling
point determination are also indispensable.
Such
characteristic nature of chemistry influences the structure of chemical
education, in particular, the structure of textbooks of chemistry.
The time allocated to chemistry teaching is necessarily limited
and hence the number of pages of chemistry textbooks is limited.
It is not an easy task to select what is to be taught and what is
to be described in the textbook.
We
presuppose, as the reader of this series, 1st-3rd university students
who have studied some chemistry in high school and who are planning
or have already decided to choose chemistry or related field as
their carrier.
This
series of textbooks, "Introduction to Chemistry" is a
proposal on the guideline of the materials to be taught to college
level students (first and second year students of university).
By editing and writing this series, we wanted to show the reader
the bridge from the basic part to the developing part. We further
attempted to the reader the goal to be attained by studying with
this series.
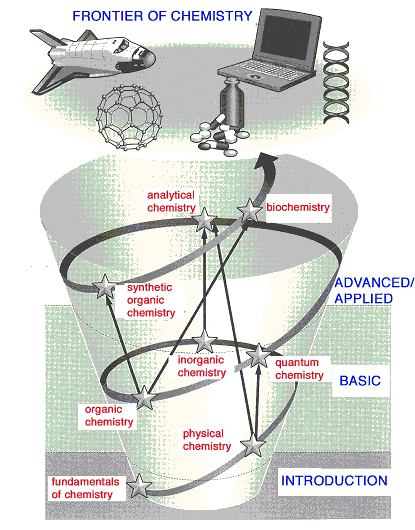
Fig. 1 Schematic Representation of the Structure of "Fundamentals
of Chemistry" Series.
The eight branches of chemistry printed in red correspond to the
titles of eight volumes of the series.
The
structure of knowledge of chemistry is "reverse-triangle",
in that the basis is more or less fixed and concrete, and not very
wide, while the upper structure is continuously developing and expanding.
The general concept of the structure of the series is depicted in
Fig. 1. The role of introductory chemistry, and hence this series,
is to indicate young students (and citizens who want to learn chemistry
which is not familiar with the subject to them) the borderline between
basic and advanced/developed area of chemistry. In other word, what
is the basic part of chemistry, and what is its developing part.
----------
LEARN
CHEMISTRY via the INTERNET
with the aid of eight volumes of top-quality textbooks in English
The series covers all branches of introductory chemistry
and are provided in pdf format.
Absolutely Free!
Q:
What are the titles of the eight volumes and who are the authors?
A: The general title of the series is: Introductory Chemistry published
by Iwanami Shoten
vol. 1 Basic Chemistry by Y. Takeuchi (ex. University of Tokyo)
vol. 2 Physical Chemistry by K. Seki (Nagoya University)
vol. 3 Inorganic Chemistry by T. Saito (ex. University of Tokyo)
vol. 4 Organic Chemistry by Y. Takeuchi (ex. University of Tokyo)
vol. 5 Synthetic Organic Chemistry by H. Suzuki (ex. Kyoto University)
vol. 6 Quantum Chemistry by K. Ohno (Tohoku University)
vol. 7 Analytical Chemistry by Y. Umezawa (University of Tokyo)
vol. 8 Biochemistry by A. Ikai (Tokyo Institute of Technology)
originally written in Japanese and now translated into English .
Q:
Why are textbooks originally written in Japanese being used for
this project?
A: There are a large number of good-quality English-language textbooks
available which cover all branches of chemistry; however, no author
or publisher will provide free public access to their textbooks
via the INERNET.
Q:
Why then are Japanese authors/publishers distributing their textbooks
free of charge?
A: There are several reasons, but the main one is that these textbooks
have sold well and the investment made in them has already been
recovered. This activity is a kind of gift back to the people from
the authors and publishers. As you know, there are many people who
cannot afford good textbooks.
Q:
Is there any other reason?
A: Providing these textbooks free of charge on the internet also
serves to promote chemistry. We are keen to get more people interested
in chemistry. If good textbooks are available for free, the opportunity
for people to get involved in chemistry will be increased.
Q:
You can obtain a lot of teaching/educational material via the INTERNET.
Is there any rationale to add another series of textbooks to the
wealth of teaching material already available?
A: Most of the information already available is supplementary material.
The limitation of supplementary material is that it is indeed ´supplementary´
and cannot substitute a good textbook. Supplementary material is
useful and only useful when you have a good textbook, without which
effective study is impossible.
Q:
Are there people who would want to use these textbook via the INTERNET?
A: If you are a student you are generally surrounded by textbooks,
and it is difficult to imagine a situation where you cannot find
a textbook on the subject you are going to study. For other members
of the public, however, such as those who have already left school/university,
textbooks are not necessarily easily accessible. Sometimes one cannot
buy textbooks due to their often high costs.
Q:
At what level are the textbooks aimed?
A: The original intention of editing this series was to provide
a series of standard textbooks for 1st and 2nd year college students
for all branches of chemistry. The textbooks will be useful even
for 3rd and 4th year students not majoring in chemistry.
Q:
How we can gain access to the textbooks?
A: Just visit the IUPAC CCE
web page [2]. There is a window for WCEN (World-wide
Chemical Education Network) [3]. There you will find "Learn
Introductory CHEMISTRY via the Internet" [1] and links to files
via each authors' individual website.
Q:
When will the textbooks become accessible via the Internet?
A: The announcement will be made via WCEN. It is expected that textbooks
will be made open one by one. Two of them are already available
in July 2004, and the remainder will be available in one or two
years.
NOTE:
This project is maintained on a 100% volunteer basis. Commercial
use of any part of the textbooks is strongly discouraged.
[1]
Learn Introductory CHEMISTRY via the Internet;
http://www.t.soka.ac.jp/chem/iwanami/index.html.
[2] IUPAC CCE web page; http://www.iupac.org/standing/cce.html.
[3] World-wide Chemical Education Network; http://www.t.soka.ac.jp/chem/wcen.html.
Last
modified
26.07.04
|









